This article originally ran in FloridaMD magazine in the November 2014 issue and was co-written by Jeffrey Phillips, MD
Obstructive sleep apnea (OSA) is a very prevalent condition. It has been estimated to occur in up to 4% of middle-aged men and 2% of middle-aged women. It is also associated with increased risk for cardiovascular morbidity/mortality. CPAP/BiPAP therapy is effective in treating OSA, but tolerance of and compliance with CPAP/BiPAP therapy is incomplete. Therefore, alternative therapies are needed for many patients with OSA.
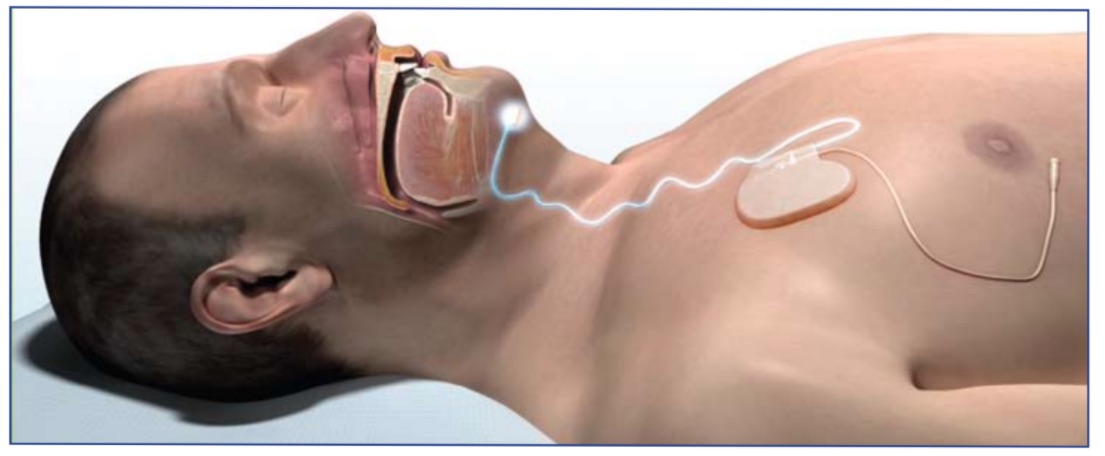
One such therapy is Inspire®, which was recently approved by the FDA to treat OSA. This involves placement of a small implanted system, which delivers mild stimulation to airway muscles to keep the airway patent during sleep. Using a small handheld remote control, the patient turns the therapy on before bed and turns the therapy off upon awakening. Inspire® therapy includes a stimulation electrode which is placed on the hypoglossal nerve to recruit tongue protrusion function. There is also a sensing lead, which is placed between the internal and external intercostal muscles to detect ventilatory effort. The neurostimulator unit is implanted in the right mid infraclavicular region. Once implanted, settings are optimized by a sleep study (polysomnography) about 4 weeks post operatively.
The STAR trial
This therapy was studied in the STAR trial (stimulation therapy for apnea reduction). The study was published in the New England Journal of Medicine in January of 2014. This study was conducted in 22 centers across the United States and Europe and was a 12-month study. The first author was Dr. Patrick Strollo from the University of Pittsburg Medical Center. The patients were predominantly men (83%), there were 126 participants with a mean age of 54.5 years. The mean body mass index (BMI) was 28.4, which is certainly lower than the typical BMI seen in patients with OSA. The patients in the study either had previous difficulty accepting or adhering to CPAP therapy. The mean apnea hypopnea index (AHI) decreased by 68% after 12 months of Inspire® therapy, from an average of 29.3, before therapy to an average of 9.0 events per hour after therapy. The oxygen desaturation index score decreased by 70% (from 25.4 desaturation events per hour down to 7.4 desaturation events per hour). The rate of procedure related serious adverse events was less than 2%. The primary outcome was based on a cohort study with participants serving as their own control.
In addition, the therapy withdrawal effect was tested in a randomized study after participants completed 12 months of follow-up. One week therapy withdrawal led to a return of OSA severity similar to baseline level without therapy. The study also demonstrated improvement in subjective measurements of the severity of OSA, such as daytime sleepiness. Subjects were excluded if their BMI was above 32 or if they had neuromuscular disease, intrinsic lung disease, pulmonary hypertension, valvular heart disease, congestive heart failure, re- cent myocardial infarction, severe cardiac arrhythmias within six months, if they had active psychiatric disease or persistent uncontrolled hypertension.
17% of the patients in the STAR study had undergone previous uvulopalatopharyngoplasty. BMI did not change on average over the 12 months of the study. The median time for surgical implantation was 140 minutes (range 65-360 minutes). The vast majority of the patients were discharged from the hospital within 24 hours of surgery. Two of the participants in the study had to have repositioning and fixation of the neurostimulator to relieve discomfort. Most of the adverse events occurred within 30 days after implantation and included sore throat from intubation, pain at the incision site and muscle soreness. 40% of participants in the study reported some discomfort associated with stimulation. The stimulation of the hypoglossal nerve is unilateral and synchronous with ventilation.
The recommendation is to screen potential participants through a drug induced sleep endoscopy procedure to help identify upper airway collapse that was likely to be focused on the retrolingual region and would therefore be amenable to forward motion of the base of the tongue by means of neurostimulation. If there is complete concentric collapse at the palate with sleep endoscopy then the patient would not be a candidate for the procedure. Patients who have more than 25% Central apneas are not felt to be candidates for Inspire® therapy at this time. The recommended age for Inspire® is 22 and above.
Compliance with the upper airway stimulation system was excellent at 86% daily use. The device was used for an average of more than five hours per night. It is worth noting that some participants (15%) had an increase in their AHI after 12 months, this was down to 7% at 18 months post-op. The STAR authors did an analysis but could not identify predictors to differentiate between those patients who responded versus non-responders.
The FDA’s approval
Based on the data from the STAR trial, the FDA approved Inspire® therapy. This therapy is only approved for a subset of patients with moderate to severe OSA (AHI 20-65/hour) who have less than 25 percent central apneas. Also, there must not be evidence of complete concentric collapse of the palate identified by a pre-op drug induced sleep endoscopy. BMI should not be above 32, so this therapy will not be appropriate for OSA patients with morbid obesity. CPAP/BiPap therapy should be offered before Inspire®, and only those patients unable to use CPAP/BiPap should undergo this surgical procedure. Snoring improved in 85 percent of patients, but Inspire is NOT intended as a primary treatment for snoring. Patients who have undergone this procedure will be unable to undergo MRI imaging. The STAR trial shows that with this careful patient selection Inspire® therapy (on average) reduces AHI significantly and improves daytime sleepiness with good compliance and an acceptable safety profile. This provides an important alternative therapy for patients with moderate-to-severe OSA who are unable to tolerate CPAP therapy.

Daniel T. Layish, MD
Pulmonologist
Dr. Layish is a Board Certified Pulmonologist and serves as the Vice President, Medical Director of Clinical Research, and Co-Director of the Adult Cystic Fibrosis Program at Central Florida Pulmonary Group in Orlando, Florida. He is also the Past-President of the Southeastern Thoracic Association and a Doctorpedia Founding Medical Partner.

Jeffrey Phillips. MD
ENT
Dr. Phillips is a leader in the treatment of snoring and sleep apnea in North and Central Florida, and is dedicated to improving sleep and overall health.
